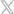Meet Anesthesia Guidelines with One Device that Provides Statistically Superior EtCO
A leading cause in preventable adverse outcomes in sedated patient cases is failure to use or correctly interpret capnography in detecting respiratory depression.2
When opiates used in sedation cause respiratory depression, the sedated patient is unable to wake themselves up from being deprived of oxygen. Respiratory depression that goes undetected can have severe effects, such as coma or death.3
Anesthesia guidelines from the American Society of Anesthesiologists, the American Association of Oral and Maxillofacial Surgeons Parameters of Care, and the American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care, require real-time end-tidal carbon dioxide (EtCO2) monitoring for all medium to deep sedation cases because capturing EtCO2 measurements effectively and accurately is critical to sedated patient safety.4-6
Not every EtCO2 monitoring cannula consistently provides both accurate EtCO2 monitoring and the necessary supplemental O2 delivery.
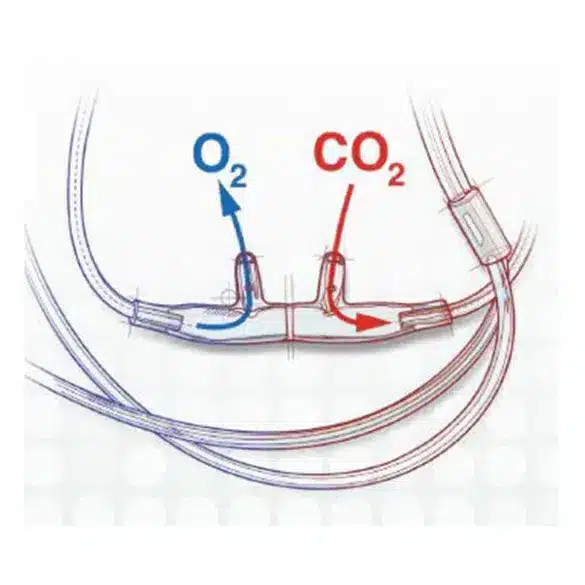 Divided Nasal Sampling Cannulas from Salter Labs Provide True EtCO2 Sampling and Effective O2 Delivery
Divided Nasal Sampling Cannulas from Salter Labs Provide True EtCO2 Sampling and Effective O2 Delivery
The divided nasal sampling cannulas from Salter Labs were designed with a divided face piece that separates O2 delivery from CO2 monitoring. These cannulas provide true EtCO2 sampling and O2 delivery without limitations at higher lpm rates.2,7
In a clinical study1 conducted by Zablocki Veterans Affairs Medical Center and Medical College of Wisconsin, the divided nasal sampling cannulas from Salter was the only nasal sampling cannula to demonstrate statistically significant superiority for both EtCO2 measurement and O2 delivery.
The divided nasal cannulas from Salter Labs feature Salter Eyes – an innovative, patented design created by anesthetists for anesthetists. These Eyes provide safety apertures in each prong to reduce the possibility of occlusions.8
To learn more about the divided nasal sampling cannula from Salter Labs, please email us or call 888-891-1200.
MED Alliance Group is a medical device distributor with more than 350 years of combined medical device sales and distribution experience. Dedicated to meeting the needs of its clinical customers and manufacturing partners, MED Alliance offers cost-effective, customized sales, logistics, and distribution solutions for products found in anesthesia/respiratory, blood/transfusion therapy, EMS/emergency room, interventional radiology/cath lab, iv/vascular and NICU/PICU.
Please follow us on LinkedIn, Facebook and Twitter for MED Alliance product updates.\
References
1 Ebert TJ, Novalija J, Uhrich TD, Barney JA. The effectiveness of oxygen delivery and reliability of carbon dioxide waveforms: a crossover comparison of 4 nasal cannulae. Anesth Analg. 2015;120(2):342-438
2 Cook TM, Woodall N, Harper J, Benger J, on behalf of the Fourth National Audit Project. Major complications of airway management in the UK: results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 2: intensive care and emergency departments. Br J Anaesth. 2011;106(5):632-642. doi:10.1093/bja/aer059
3 https://www.medicalnewstoday.com/articles/319030.php
4 Standards for basic anesthetic monitoring. American Society of Anesthesiologists. 2011.
5 Parameters of Care: Clinical Practice Guidelines for Oral and Maxillofacial Surgery. American Association of Oral and Maxillofacial Surgeons. 2012.
6 Neumar RW, Otto CW, Link MS, et al. Part 8: Adult Advanced Cardiovascular Life Support. 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122(suppl 3):S729-S767.
7 Roth JV, Barth LJ, Womack LH, Morgenlander LE. Evaluation of two commercially available carbon dioxide sampling nasal cannulae. J Clin Monit. 1994;10(4):237-243
8 Salter Labs Technical Report 006