ED Clinicians: What’s an Easy Option When Oral or IV Access is Not?
 Patients who come into the Emergency Department can sometimes be so disoriented that taking oral medication is not possible. Clinicians often struggle to obtain IV access to administer medications or fluids when a patient is extremely dehydrated or their extremities are severely burned or injured. In such situations, the Macy Catheter can be an easy alternative, especially when central line placement is not necessary.
Patients who come into the Emergency Department can sometimes be so disoriented that taking oral medication is not possible. Clinicians often struggle to obtain IV access to administer medications or fluids when a patient is extremely dehydrated or their extremities are severely burned or injured. In such situations, the Macy Catheter can be an easy alternative, especially when central line placement is not necessary.
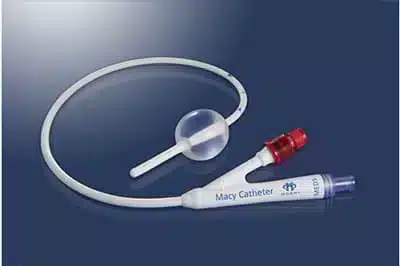
The Macy Catheter is a rectal catheter that provides a quick and easy route for medication and fluid delivery to help facilitate rapid symptom management. The thin silicone catheter administers medication via rectal mucosal absorption.
According to the Case Report, “An alternative for rapid administration of medication and fluids in the emergency setting using a novel device,” published in the American Journal of Emergency Medicine, “Placement is easy and fast, with no need for sterility or advanced training.”
The Report also states, “Absorption through rectal mucosa is rapid, with blood levels of many drugs (aqueous and alcoholic solutions in particular) becoming therapeutic very rapidly.” Clinicians involved in the case study noticed the medication effects soon after administration.
Medications delivered via the Macy Catheter partially avoid the first pass of the liver metabolism, making medication blood serum concentration rise more rapidly compared to the oral route.
Clinicians reported being appreciative of not having to make multiple attempts for vascular access.
The Macy Catheter enables both clinicians and patient caregivers to administer medication repeatedly without invading the patient’s privacy each time medication is needed. The catheter can stay in place for 28 days.
Based in Newark, California, Hospi Corporation was founded in 2012 and is dedicated to improving quality of life by innovating practical, high impact medical devices that enhance patient comfort and wellbeing, ease caregiver burden, and reduce cost. The Macy Catheter received 510(k) clearance from the U.S. Food and Drug Administration in 2014.
For more information about the Macy Catheter, contact your local MED Alliance Group Sales Representative, call 888-891-1200 or email us.