Study Shows Catheter-Related Bloodstream Infections Increase Three-Fold After Dressing Disruption
Preventing early dressing changes to decrease the risk of CLABSI
Each year, approximately 250,000 cases of bloodstream infections occur hospital-wide.1 According to an article in the Journal of Healthcare Contracting1, one of the main causes of central line-associated bloodstream infections (CLABSI) is an infection at the exit site due to patient exposure to the environment or caregiver, such as moisture from bathing.
A catheter-related infection study2 of 1,419 patients found
- 296 colonized catheters
- 29 major catheter-related infections
- 23 catheter-related bloodstream infections
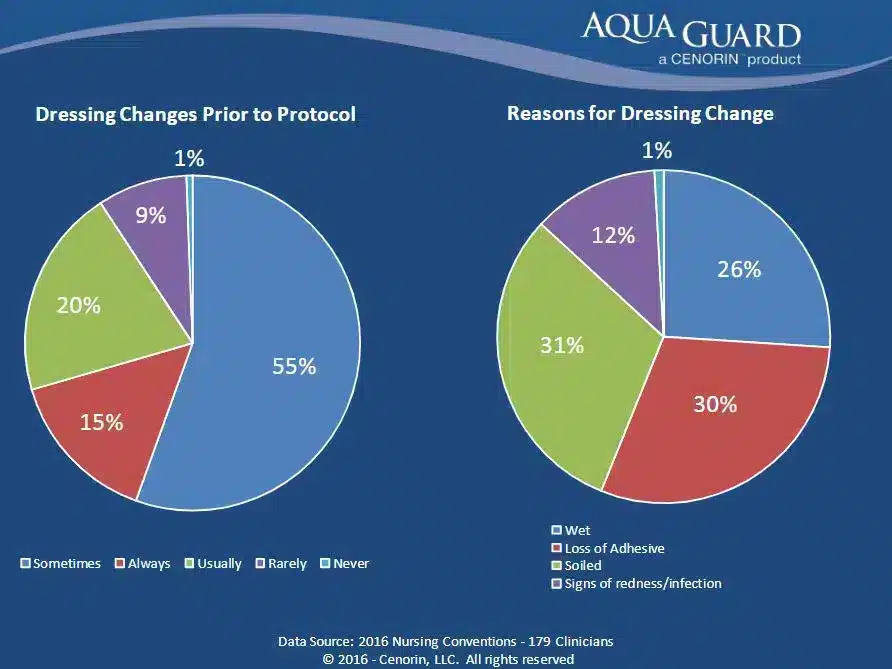
Sixty-seven percent of the 11,036 dressing changes were performed before the planned date due to soiling or undressing. The study showed that the “risk of major catheter-related infection and catheter-related bloodstream infection increased by more than three-fold after the second dressing disruption and by more than ten-fold if the final dressing was disrupted.”
CLABSIs cost approximately $46,000 per case – and even more importantly, most cases are preventable with proper sterile techniques, management strategies and close observation. With each PICC-related (peripherally inserted central catheter) blood stream infection, patient hospital stay can be prolonged, and the risk of death can increase.
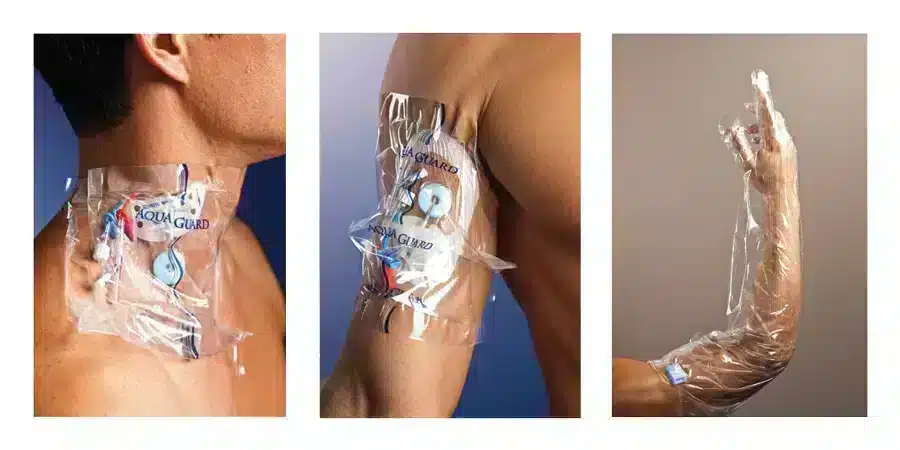
Dressing changes are important to keep the site dry and clean, but there are devices on the market to help protect dressings from environmental factors that cause early changes, such as moisture.
IMPROVE QUALITY OF CARE WITH AQUAGUARD
AquaGuard moisture barriers from TIDI were designed to help protect catheter sites, surgical incisions or dressings during patient showering. This one-time use barrier helps reduce infections of wound and IV sites. AquaGuard can help reduce nursing time and expensive supply costs by eliminating the need to reapply dressings after patient showering.
AquaGuard is latex and DEHP-free and utilizes a gentle, medical-grade adhesive for minimal patient discomfort upon removal.
To learn more about AquaGuard, please email us or call 888-899-1200.
MED Alliance Group is a medical device distributor with more than 350 years of combined medical device sales and distribution experience. Dedicated to meeting the needs of its clinical customers and manufacturing partners, MED Alliance offers cost-effective, customized sales, logistics, and distribution solutions for products found in anesthesia/respiratory, blood/transfusion therapy, EMS/emergency room, interventional radiology/cath lab, iv/vascular and NICU/PICU.
Please follow us on LinkedIn, Twitter and Facebook for MED Alliance product updates.
References