Cath Lab Physicians and Staff Can Decrease Their Radiation Exposure Risk by Doing this One Thing
According to Cardiovascular Diagnosis and Therapy, over one million cardiac catheterization procedures are performed annually in the United States.3 Although cardiac catheterizations are generally considered safe, radiation exposure is a risk for the patient as well as the interventional cardiologists and cath lab personnel.
Over the past decade, older cardiac interventionalists have shown high rates of radiation complications, such as cataracts and left-side brain cancers.1 Many of these cases are thought to be linked to the use of ionizing radiation.
RISK TO INTERVENTIONAL CARDIOLOGISTS
A 2013 study found that interventional cardiologists have a two to 10 times higher radiation exposure rate than diagnostic radiologists. According to the study, “Cumulative doses after 30 years of working life are in the range of 50 to 200 [millisieverts] mSv, with a projected professional lifetime attributable excess cancer risk in the order of magnitude of 1 in 100.” The study also concluded that the left side of the operator is 30 to 100 percent more exposed than the rest of the body. Less protected parts of the body such as the operator’s head and hands can receive doses between 5 to 50 mSv per year.4
RISK TO CATH LAB STAFF
A study focusing on radiation exposure of cath lab technicians and nurses suggested that these staff members may benefit from additional protection. In this two-phase study, 401 cath lab staff received standard radiation protection, while 363 received the same protection with the addition of an accessory lead shield placed between the patient and the staff member. The study concluded that radiation exposure to the staffer was decreased by nearly two-thirds when the accessory lead shield was used.5
IMPROVE CATH LAB PERSONNEL PROTECTION WITH RADPAD®
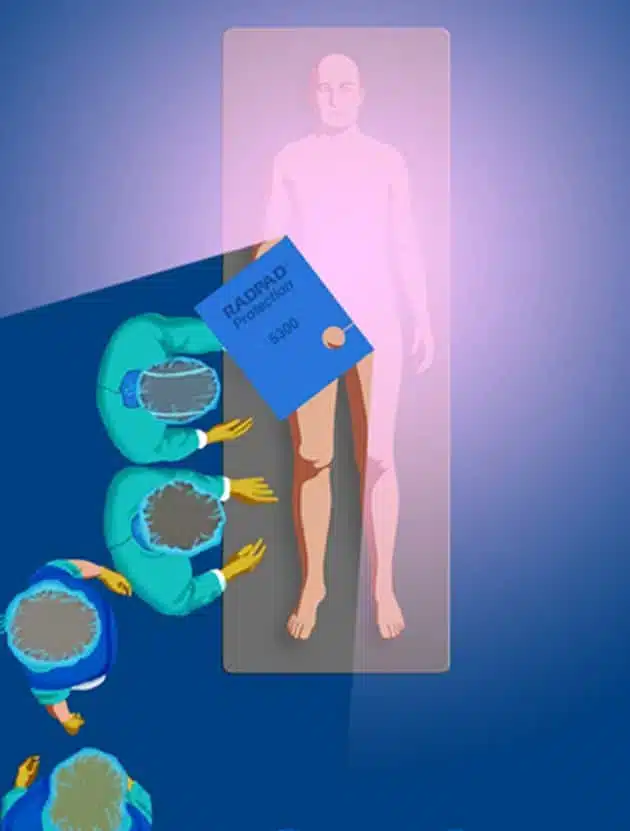
The RADPAD® is the only clinically-proven, non-lead and non-vinyl, lightweight and disposable shield that can guarantee a 95% reduction in x-ray scatter radiation. The RADPAD® shield works by creating a “shade zone” on the patient where radiation is absorbed into the shield. This allows the clinician to move freely within this zone with significantly reduced scatter radiation, all while protecting the patient.
RADPAD® 5000 series products are comprised of several procedure-specific radiation protection shields designed to provide maximum protection to the operator and cath lab personnel during fluoro-guided procedures.
RADPAD® 7000 series are comprised of several procedures specific sterile drape + RADPAD® Radiation Protection Shields, designed to protect operators and cath lab personnel during fluoro-guided procedures.
RADPAD® No Brainer® is an attenuation material-lined scrub cap worn by the cath lab personnel to protect their brain from scatter radiation during fluoro-guided procedures.
RADPAD® Thyroid Shield w/ Cover is a RADPAD® thyroid shield worn by the cath lab personnel to protect thyroid glands during fluoro-guided procedures.
To learn more about the RADPAD® and how your cath lab personnel can stay protected, please email us or call 888-891-1200.
MED Alliance Group is a medical device distributor with more than 350 years of combined medical device sales and distribution experience. Dedicated to meeting the needs of its clinical customers and manufacturing partners, MED Alliance offers cost-effective, customized sales, logistics, and distribution solutions for products found in anesthesia/respiratory, blood/transfusion therapy, EMS/emergency room, interventional radiology/cath lab, iv/vascular and NICU/PICU.
Please follow us on LinkedIn, Facebook and Twitter for MED Alliance product updates.
References
1 Defining the Cath Lab Workplace Radiation Safety Hazard _ DAIC
2 https://www.goodsamaritanmc.com/our-services/heart-vascular/cardiac-cath-lab




