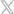Pulmodyne® O2-MAX System –Integrated Intake Filter to Protect Against COVID-19
As COVID-19 continues to see surges across the United States, effective and safe respiratory care is especially important for both the patients and clinicians. Pulmodyne®, an ISO certified manufacturer of respiratory devices, has fulfilled this need with the O2-MAX™ System with Integrated Nebulization.
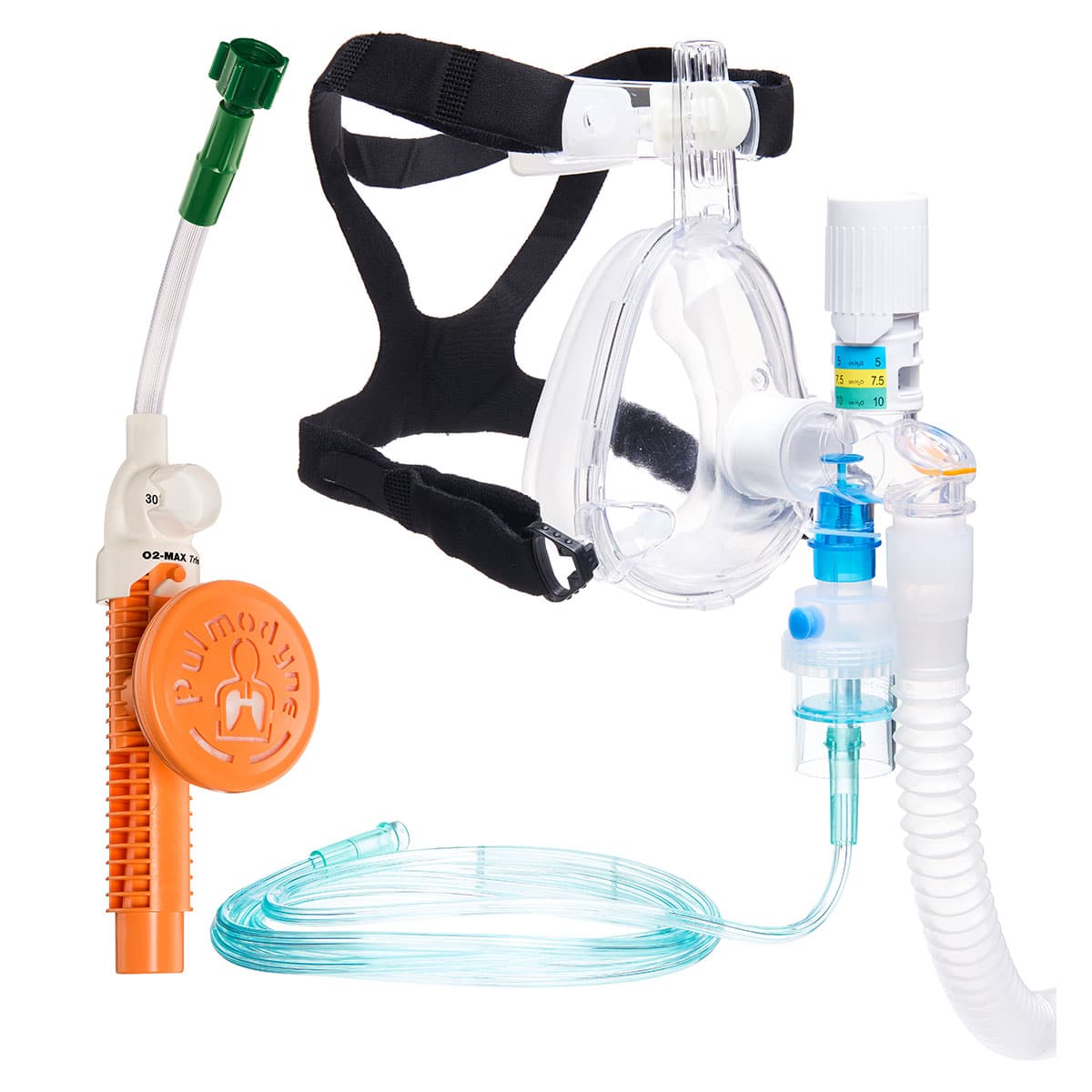 The O2-MAX™ features an intake filter with bacterial and viral protection to patients. The viral efficiency of the O2-MAX filter is 99.999%, which can help protect against the COVID-19 virus. Since March, Pulmodyne® has been in full overtime production to fulfill the increased demand for respiratory devices capable of protecting against COVID-19.
The O2-MAX™ features an intake filter with bacterial and viral protection to patients. The viral efficiency of the O2-MAX filter is 99.999%, which can help protect against the COVID-19 virus. Since March, Pulmodyne® has been in full overtime production to fulfill the increased demand for respiratory devices capable of protecting against COVID-19.
In addition to the highly effective intake filter, the O2-MAX™ allows healthcare providers to easily provide airway management from one device due to the integrated closed-circuit nebulization feature that does not interfere with the patient’s CPAP therapy.
Other features of the O2-MAX™ include:
- Delivery of up to 140 LPM of flow with an approximate 30% FiO2
- A 3-SET™ option for PEEP of 5, 7.5 and 10 cm H2O, and 5-SET™ option of 5, 7.5, 10, 12.5 and 15
- Integrated nebulization without interrupting the delivered flow to the patient
To learn more about the O2-MAX™ with Integrated Nebulizer from Pulmodyne, call 888-891-1200 or email us to be connected to your local representative.
MED Alliance Group is a medical device distributor that has been dedicated to meeting the needs of our clinical customers and manufacturing partners since 1998. We specialize in the sales, marketing, importation, logistics and distribution of innovative, high-quality and cost-effective products found in anesthesia and respiratory, blood and transfusion therapy, EMS and emergency room, interventional radiology and cath lab, iv and vascular, as well as NICU and PICU.
Please follow us on LinkedIn, Facebook and Twitter for MED Alliance product updates.